 
Each kernel is simultaneously attracted by a number of mobile electrons and each mobile electron is attracted by a number of metal ions. Therefore covalent radii are always smaller than van der waal radius.Ī metal lattice or crystal consists of positive kernels or metal ions arranged in a definite pattern in a sea of mobile valence electrons. Van der waals radius of an element is always larger than its covalent radius because:ġ)Since the van der waal forces of attraction are weak ,therefore, the internuclear distance in case of atoms held by van der waal forces are much larger than those between covalently bonded atoms.Ģ)Since a covalent bond is formed by overlap of two half filled atomic orbitals, a part of electron cloud become common. The inter nuclear distance between two adjacent hydrogen atoms of the two neighbouring molecules in the solid state 240 pm. 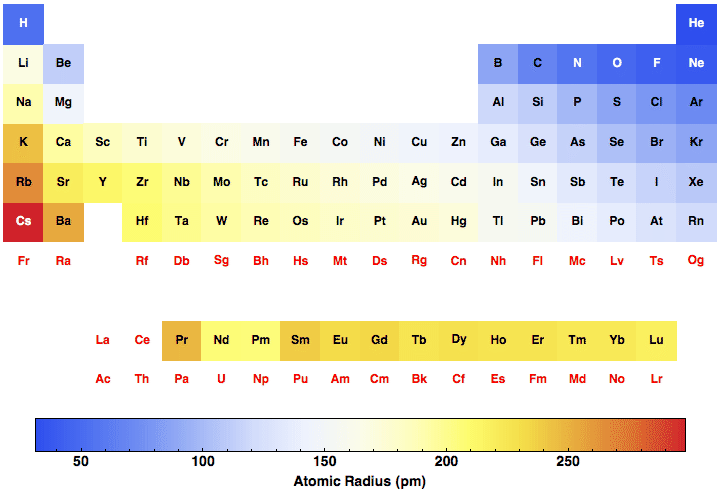
The van der waals radius of chlorine atom is 180 pm. The magnitude of the van der waal radius depends upon the packing of the atoms when the element is in the solid state.įor Example: The inter nuclear distance between two adjacent chlorine atoms of the two neighbouring molecules in the solid state is 360 pm. It Is defined as one half the distance between the nuclei of two identical non bonded isolated atoms or two adjacent identical atoms belonging to two neighbouring molecules of an element in the solid state. Since the inter nuclear distance between two bonded atoms is called the bond length. Therefore, R covalent = ½ (internuclear distance between two bonded atoms) It is defined as one half the distance between the nuclei of two covalently bonded atoms of the same element in a molecule. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
